http://www.johnbright.conwy.sch.uk/depts/science/Y13/P-Y13_Physics_Revision_Notes.html
Practical investigation
We introduced the practical investigation coursework today. Your job is to decide what areas you might like to look at. For some ideas please see http://www.nuffieldfoundation.org/practical-physics.
Assessment
When we are back after the holiday it will be doing a bit of an assessment to see how we are all getting on and what we need to revisit. The key topics are:
Ideal gases
Specific heat capacity
Boltzmann and his factor
Flux and flux linkage
Wave/particles
Momentum
Mass defect
Risks and dose
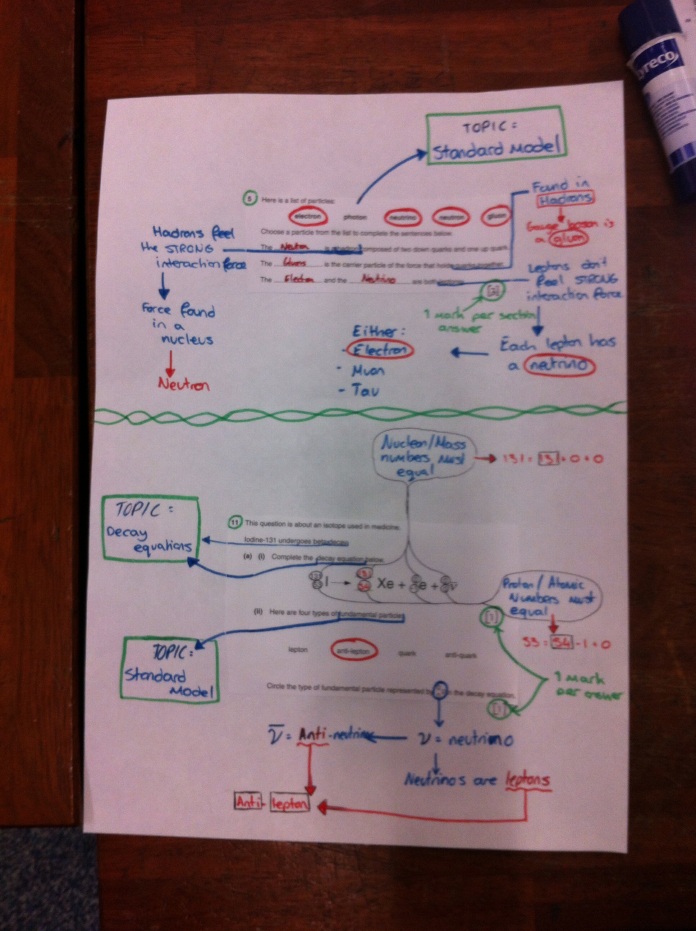
Particle names
Get revising.
Decay chains
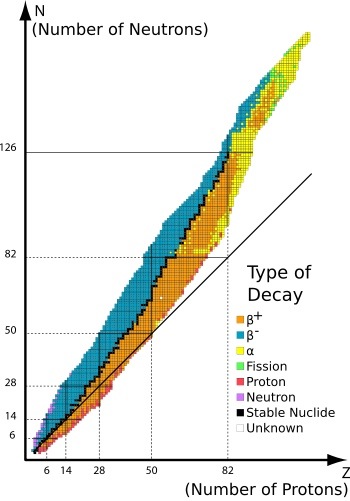
Right. A nucleus is a balance between repulsion from all those positive protons and the strong nuclear force trying to hold the whole situation together. Too many protons or too many neutrons leads to unbalance, instability and radioactive decay. Big elements will keep on decaying until they reach a stable element (probably lead).
We often represent this on an N-Z plot that shows what kind of unstableness leads to what decay.
http://www.a-levelphysicstutor.com/nucphys-NZ-curve.php
You need to know how each type of decay moves an atom in the plot (ideally towards the VALLY OF STABILITY).
http://www.khanacademy.org/science/chemistry/radioactive-decay/v/types-of-decay.
On an unrelated note gases can be heavy: http://www.youtube.com/watch?v=QLrofyj6a2s
Revision guide
A decent A2 revision guide can be found at http://www.amazon.co.uk/A2-Level-Physics-OCR-Revision-Guide/dp/1847622720 at a cost of £7.69. You can get it through the school for £5 plus postage.
Fission and fusion
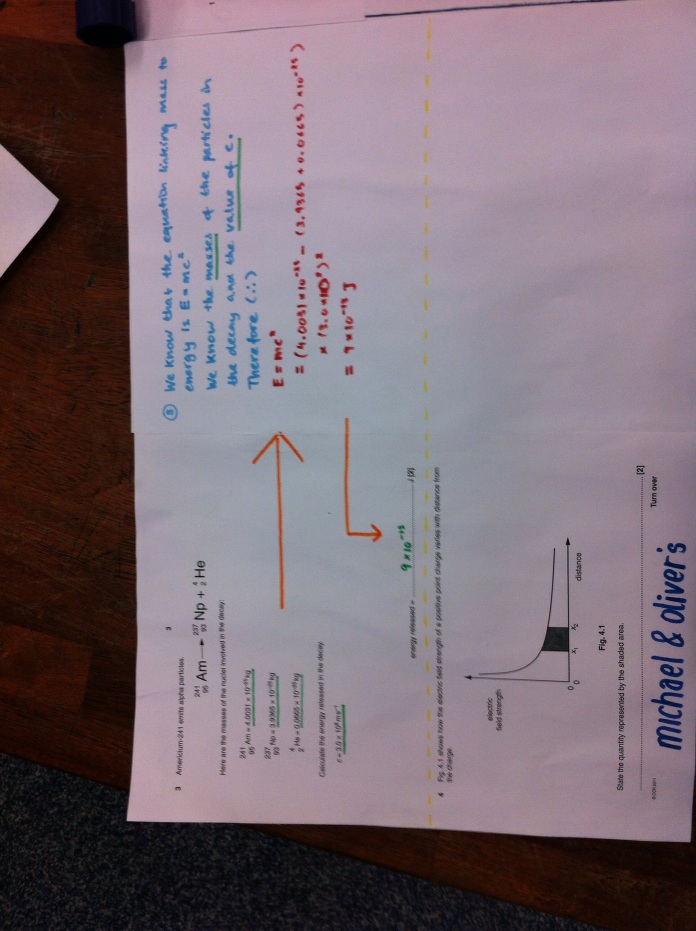
Fusion is the combining of small nuclei; fission is the breaking apart of large ones. This leads to a tiny amount of mass going missing (the mass defect) which is converted into energy and represents the binding energy of the atom.
http://www.diffen.com/difference/Nuclear_Fission_vs_Nuclear_Fusion
http://physics.stackexchange.com/questions/32699/how-to-explain-e-mc2-mass-defect-in-fission-fusion
http://hyperphysics.phy-astr.gsu.edu/hbase/nucene/nucbin.html
Dose
When absorbing radiation you have to be careful. Too high an energy focused on too small an area can prove dangerous. Dose and dose equivalent are good ways of measuring this. Here the Canadians are explains it: http://www.cnsc-ccsn.gc.ca/eng/readingroom/radiation/radiation_doses.cfm. Effective dose is interesting but not part of our course. As a useful rule of thumb a dose of 1 Sv increases your chance of cancer by 3%.
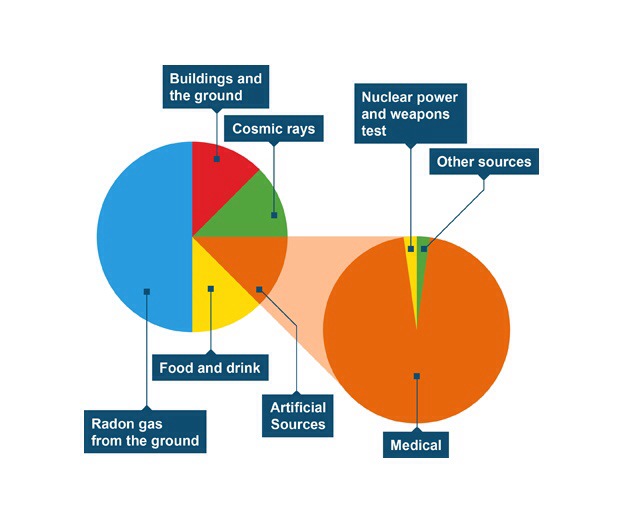
Remember Banbury is one of the most radioactive places in the UK; it’s the radon.
Picturing relativity
Not at all relevant to what we are currently doing but good none the less http://m.youtube.com/watch?v=C2VMO7pcWhg&desktop_uri=%2Fwatch%3Fv%3DC2VMO7pcWhg
Exam questions answered
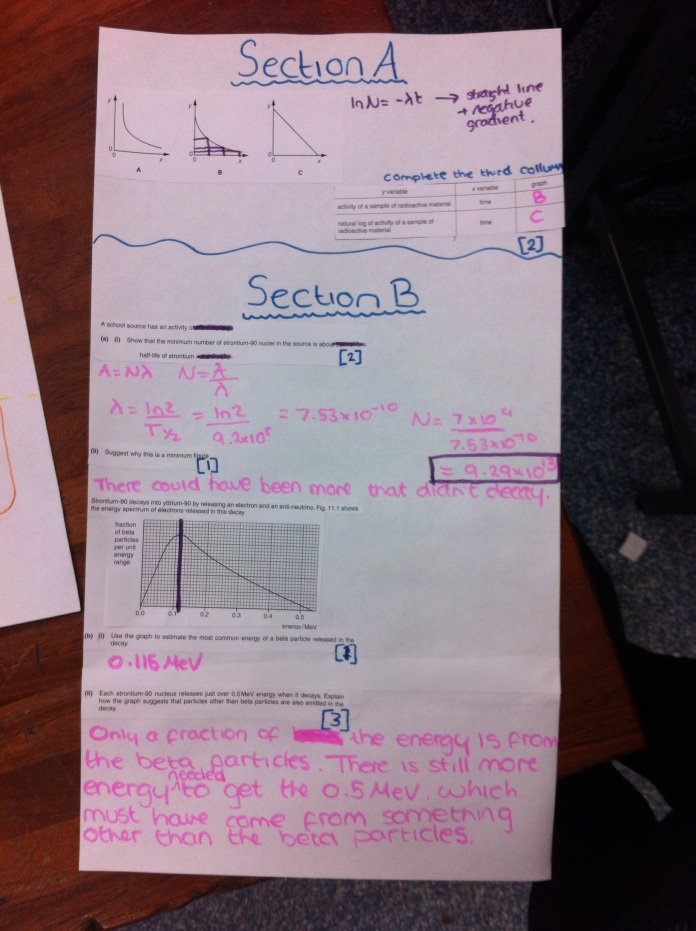
Here are the questions you have answered. The key to this is the explanations that go alongside this to help you with the questions.
Rotate head as needed.